Where pharmaceutical decisions break down
Structural friction emerges when regulatory, commercial, and governance pressures converge.
Cross-Functional Misalignment
Regulatory, legal, medical, and commercial functions often operate in parallel—without a unified decision framework.
Compressed commercial timelines
Launch pressure accelerates decisions before regulatory and governance implications are fully mapped.
Data without decision context
Information is abundant—but its impact on exposure, precedent, and long-term risk is rarely structured for decision-making.
Governance and accountability gaps
Decisions move forward operationally while defensibility and board-level visibility lag behind execution.
What CSuitePharma is
What it's not
What is not
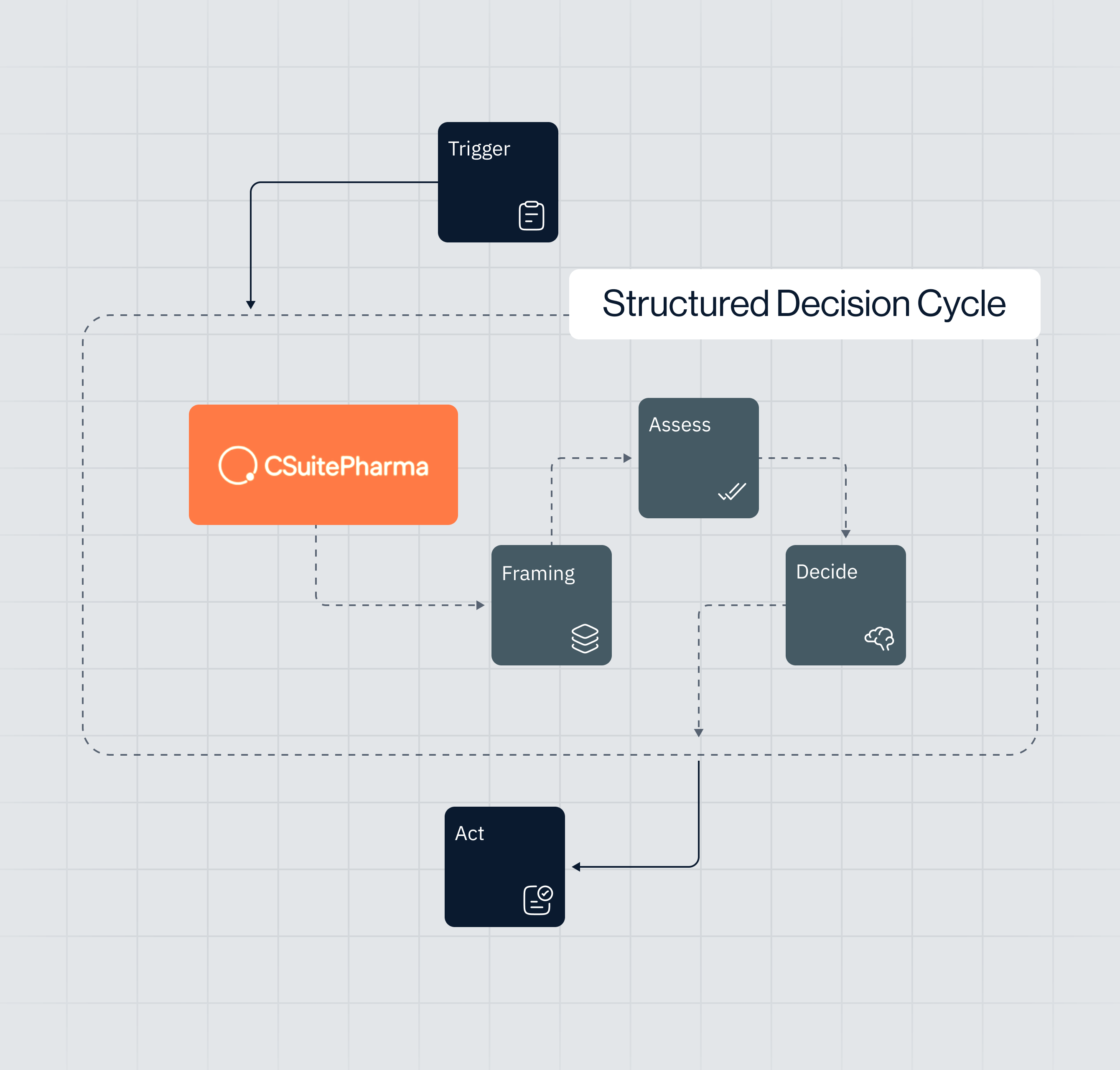
Inside the CSuitePharma decision environment
Structured evaluation of material pharmaceutical decisions
Grounded in regulatory, governance, and commercial realities.
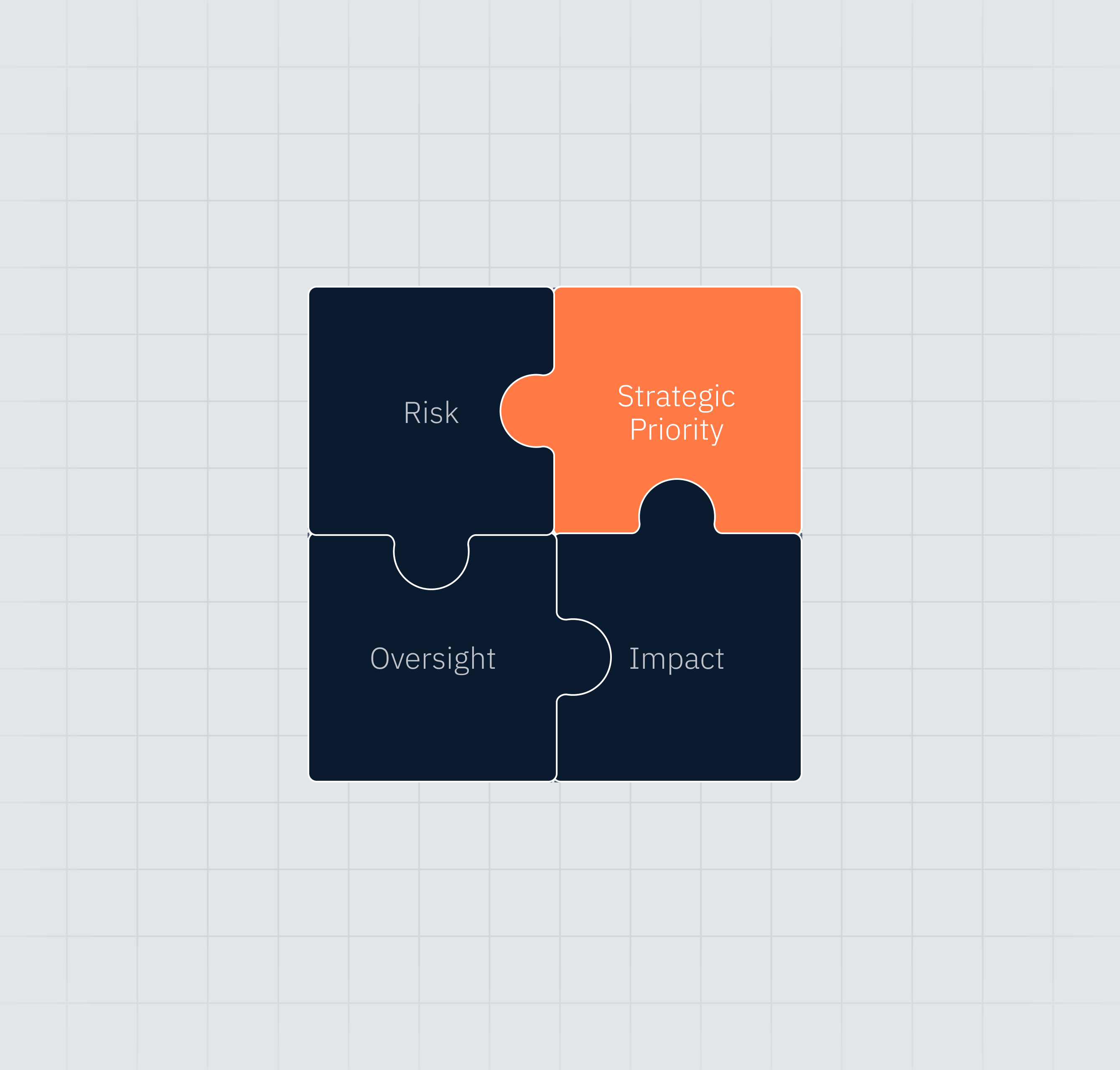
Structured evaluation of strategic tradeoffs
Risk, oversight, impact, and priority evaluated together—not in isolation.
Structured pathways for defensible execution
Sequencing decisions across governance, oversight, and operational accountability.
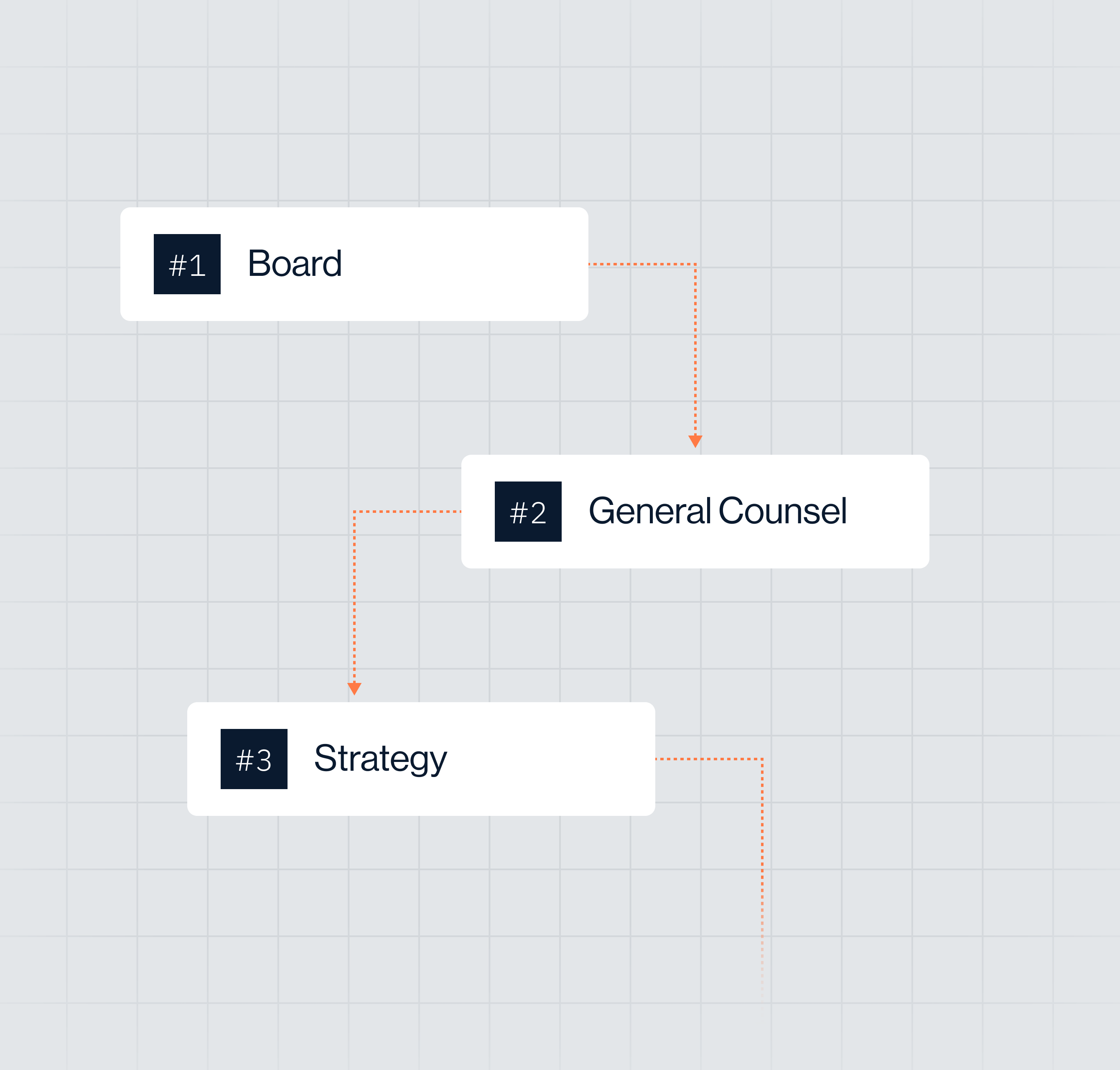
Decisions organized by executive mandate
Aligned for board, general counsel, strategy, and regulatory leadership.
Structured for Pharmaceutical Executive Leadership
Access organized by executive role and governance responsibility.
Structured evaluation of legal exposure and governance implications
Regulatory risk, oversight accountability, and cross-functional impact assessed in one integrated frame.

Structured evaluation of legal exposure and governance implications
Regulatory risk, oversight accountability, and cross-functional impact assessed in one integrated frame.

Structured evaluation of legal exposure and governance implications
Regulatory risk, oversight accountability, and cross-functional impact assessed in one integrated frame.

Structured evaluation of legal exposure and governance implications
Regulatory risk, oversight accountability, and cross-functional impact assessed in one integrated frame.

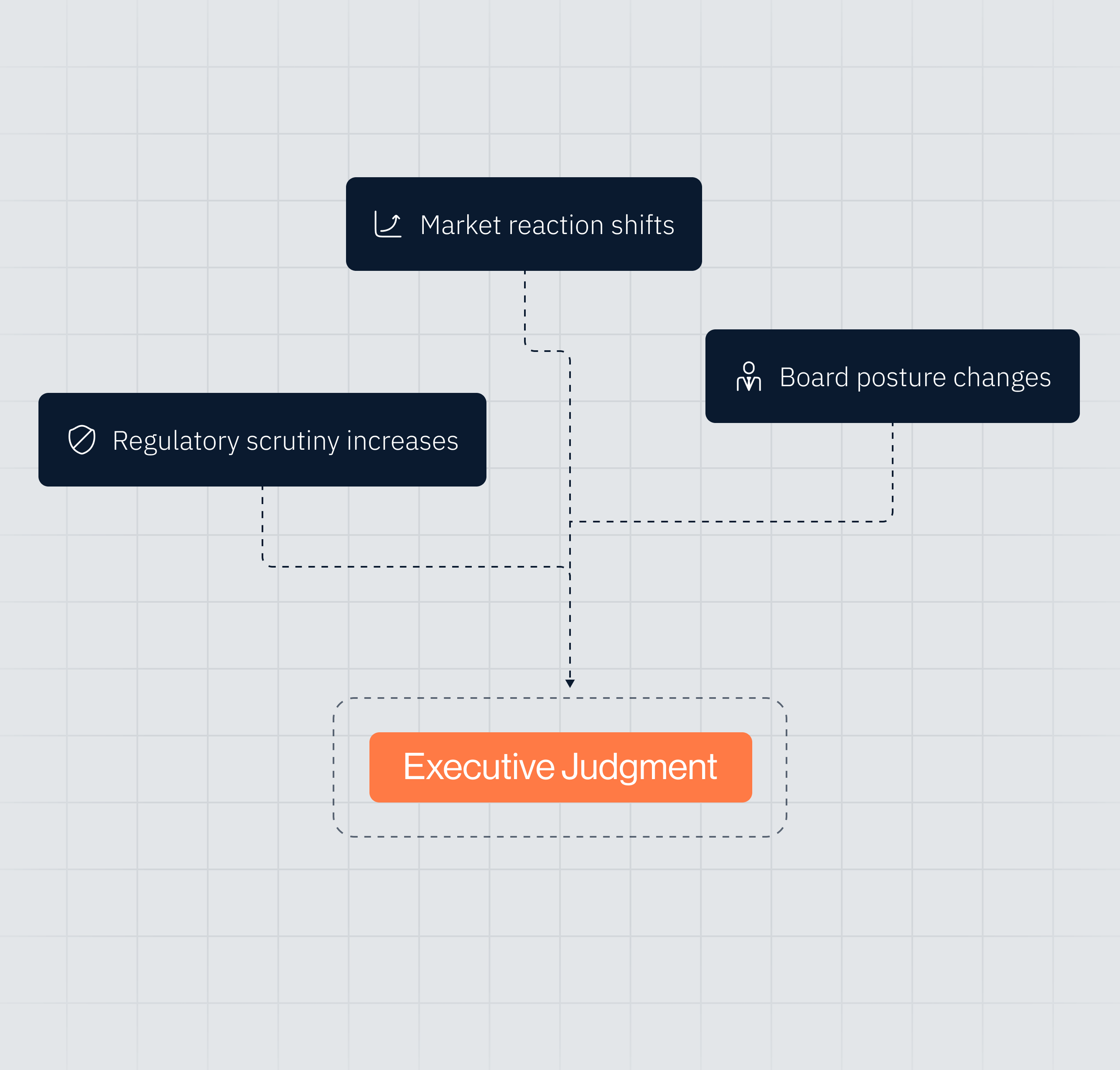
Launch Acceleration vs. Governance Exposure
Structured evaluation of litigation probability bands, regulatory certainty classifications, governance escalation thresholds, and conditional execution pathways.
Index
Pathway
Developed Within Regulated
Pharmaceutical Leadership
CSuitePharma is structured by executives who have operated inside litigation exposure, regulatory scrutiny, board-level accountability, and disclosure-sensitive decision-making within pharmaceutical organizations. Its architecture reflects enterprise governance realities — not theoretical models or automated analysis.
Built for Defensibility — Not Summary
CSuitePharma does not aggregate information. It structures decisions within governance, regulatory, and board accountability frameworks.
Exposure band classification
Structured probability tiers that define litigation and enforcement risk before action is taken.
Regulatory certainty indexing
Clear certainty gradations distinguishing validated pathways from conditional or contested routes.
Escalation trigger modeling
Predefined governance thresholds that determine when issues move to executive or board review.
Role-specific accountability mapping
Explicit assignment of oversight responsibility aligned to enterprise reporting structures.
Conditional execution pathways
Sequenced action models that adjust acceleration based on exposure and governance posture.
